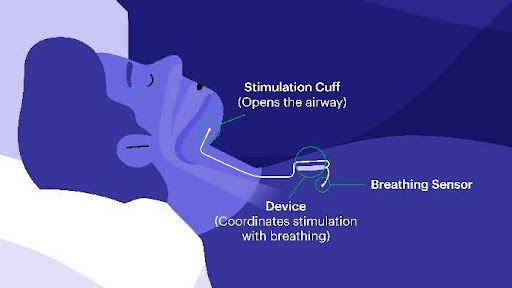
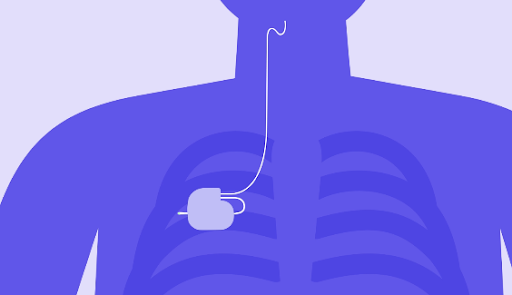
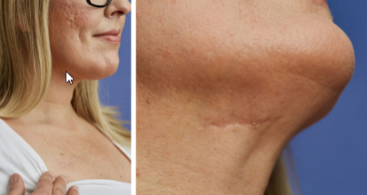
Benefits
Inspire therapy has been demonstrated to significantly reduce the severity of obstructive sleep apnea (OSA). Proper treatment may lead to an increase in the ability to perform daily tasks and a decrease in risks associated with untreated OSA: accidents (e.g., motor vehicle accidents), hypertension, stroke, diabetes, vascular disease, heart failure, and early mortality.
Potential Health Risks
- Infection
- Tongue Weakness
- Swallowing or Speech Related
- Insomnia/Arousal
- Revision Intervention
- Discomfort
- Tongue Abrasion
Activities to Avoid:
Component manipulation: Do not manipulate (move) or rub your stimulator or leads through your skin. This could lead to dislodgement, damage to the components, skin damage or unintended stimulation.
Scuba diving or hyperbaric chambers: Do not dive below 30 meters (100 feet) of water or enter hyperbaric chambers above 4.0 atmospheres absolute.